|
Respiratory syncytial virus (RSV) is a viral pathogen known for causing respiratory infections, predominantly affecting the lungs and airways. It often causes a mild cough and cold but, in some people, can trigger pneumonia or bronchiolitis, characterised by inflammation in the airways. RSV is very common. Globally, it infects up to 90% of children within the first 2 years of life and frequently reinfects older children and adults. These are more common during winter, especially in temperate countries like the UK. According to a recent study, there are roughly 33 million cases, 3.6 million hospital admissions, and 100,000 deaths globally each year in children under 5 years old. It is the second largest infectious cause of death in children under one year of age, after malaria. Despite developing antibodies after an infection, individuals remain vulnerable to RSV throughout life. Infants under 6 months old, people with weak immune systems, and the elderly are at the highest risk, with severe cases potentially leading to hospitalisation. Adult infections are often undiagnosed, particularly in the elderly, leading to an underestimated number of cases. 
|
Respiratory syncytial virus (RSV)
Infectious Disease
|
In healthy adults, RSV infection typically develops with cold-like symptoms such as runny nose, sneezing, nasal congestion, cough, and sometimes fever. In children, it can also lead to ear infections and croup, characterized by a barking cough due to upper airway inflammation. Bronchiolitis, affecting the small airways in the lungs, is a common and serious consequence in babies and infants, making breathing difficult and feeding challenging. Children under one year old, the elderly, and those with weak immune systems are at increased risk of severe RSV symptoms. RSV infection can cause pneumonia or even death.
|
|
In temperate regions like the UK, RSV recurs each year. Epidemics typically begin in October and last for 4 to 5 months, reaching their peak in December. 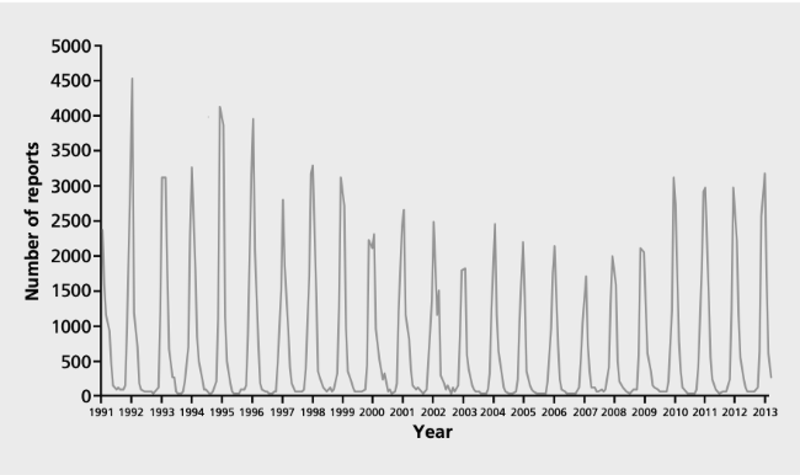
Laboratory reports of infections due to respiratory syncytial virus, England and Wales by date of report 1991-2013
Source: The Green Book
RSV spreads when someone who's infected coughs or sneezes, releasing large droplets that carry the virus. It can also be caught by touching things that have the virus on them, like door handles or toys, and then touching the face. Once exposed to the virus, it takes about 3 to 5 days for symptoms to begin. RSV transmission can be reduced by thorough handwashing and keeping surfaces that are often touched clean. It is usually best for people with a cold, or other respiratory illnesses to avoid contact with those at risk where possible.
|
|
Currently, the primary approach to RSV care is supportive, involving measures like administering fluids and oxygen as necessary. Although there's one licensed antiviral drug for RSV, its usage is uncommon due to concerns regarding drug toxicity and limited efficacy. In severe cases, intensive care may be required, including artificial ventilation for mechanical breathing support. Tragically, almost all infant fatalities from RSV occur in developing countries where access to such costly care is limited. Passive immunisation, using a monoclonal antibody (mAb) called Palivizumab that mimics the immune system’s ability to fight off RSV, is available to protect very high risk infants for whom RSV infection is likely to cause serious illness or death, such as those with severe underlying lung or heart disease, and all children less than 24 months of age with severe combined immunodeficiency syndrome (SCID). A new mAb, called nirsevimab, that could be used widely has recently been licensed alongside an RSV vaccine for pregnant women, to protect their infants and older adults. The Joint Committee on Vaccination and Immunisation (JCVI) advised that an RSV immunisation programme, that is cost-effective, is possible for both infants and older adults and the process for deciding which product to use is underway with an outcome expected in Summer 2024. |
|
This report by BBC South today from August 2017 looks at the impact that an RSV infection can have on an otherwise healthy infant. It also looks at a study being conducted by Oxford Vaccine Group and OSPREA that looks to reduce infant RSV infections by vaccinating pregnant women. Understanding RSV, a major cause of respiratory illness in infants.
https://www.youtube.com/embed/1Q7_vEwoKrI?wmode=opaque&controls=&rel=0
|
JCVI recommendation for RSV vaccination programmeThe JCVI issued early advice on June 22, 2023, advising the Department of Health and Social Care to develop a policy on RSV interventions, allowing sufficient time for the necessary planning and procurement to allow an immunisation programme to go ahead. Surveillance is also needed to monitor immunisation uptake and program impact. The JCVI has not yet expressed a preference for products as their efficacy is broadly comparable and there are no head-to-head studies to allow direct comparison. Programme to protect neonates and infants The JCVI note that a seasonal programme, seasonal programme with catch-up, or year-round passive immunity programme using monoclonal antibodies for new-borns could be cost-effective. They also note that a seasonal or year-round maternal active immunisation programme could be cost-effective. The JCVI advises a preference for a year-round offer for a passive immunisation or maternal immunisation programme to ensure high uptake and for reasons of operational effectiveness because this would be less complex and resource-intensive to deliver, compared with running seasonal campaigns. Programme for older adults The JCVI notes that an RSV vaccine programme for adults aged 75 years and above could be cost-effective, stating that this would be influenced by multi-year protection from a single dose. The JCVI advises a programme for older adults aged 75 years old and above, currently favouring a one-off campaign as the strategy for this programme with the initial offer covering several age cohorts and then a routine programme for those turning 75 years old. The delivery and implementation are to be determined through further consultation between NHS England, DHSC, UKHSA and the devolved administrations. |
Page last updated Tuesday, April 9, 2024



